
The primary outcome for this analysis was the net clinical outcome (NCO), a composite of stroke/systemic embolism events, major bleeding, or death. Methods ENGAGE AF-TIMI 48 was a multicenter randomized, double-blind, controlled trial in 21,105 patients with atrial fibrillation (AF) within 12 months and CHADS2 score >2 randomized to higher-dose edoxaban regimen (HDER) 60 mg/reduced 30 mg, lower-dose edoxaban regimen (LDER) 30 mg/reduced 15 mg, or warfarin and followed for 2.8 years (median). 
Food may have a modest effect on the pharmacokinetic properties of edoxaban.īackground: To compare the efficacy and safety of edoxaban vs. The 2 products of edoxaban tablets are bioequivalent and safe in healthy Chinese volunteers. Both formulations displayed comparable safety profiles, with no serious adverse events reported. 
Food increased exposure and prolonged the time to maximum concentration of edoxaban. The geometric least‐squares mean ratios for Cmax, AUC from time 0 to the last measurable time point (AUC0‐t), and AUC from time 0 extrapolated to infinity (AUC0‐∞) were 97.0%, 95.4%, and 96.1%, respectively, in the fasting test, and 98.6%, 100.0%, and 99.8%, respectively, in the fed test. The pharmacokinetic parameters maximum concentration (Cmax) and area under the concentration‐time curve (AUC) were compared to assess bioequivalence. Plasma edoxaban concentrations were determined using liquid chromatography–tandem mass spectrometry. All subjects randomly received 60‐mg generic (test) or branded (reference) edoxaban tablet in each period. Sixty‐four healthy Chinese subjects participated in this open‐label, randomized, 2‐sequence, 4‐period, crossover study. The aim was to investigate the bioequivalence of 2 orally administered edoxaban 60‐mg tablets and the food effects on the pharmacokinetics of edoxaban. 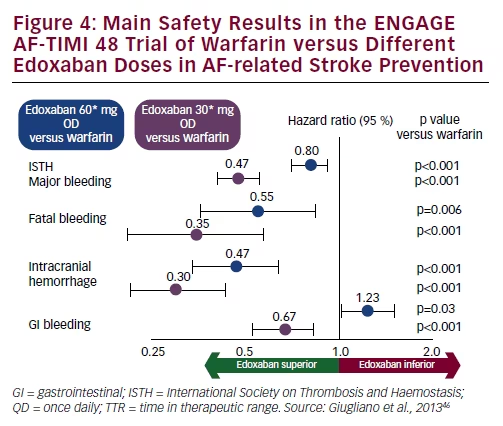
While the relative risk reductions remain similar, edoxaban provides incrementally larger absolute reductions in outcomes over warfarin in patients with higher CHA2DS2VASc scores. The benefit and safety of edoxaban versus warfarin is maintained across CHA2DS2VASc scores. Because of higher event rates the number of events prevented with edoxaban tended to be greater in patients with higher CHA2DS2VASc scores. Edoxaban reduced SSE, MB, ICH, and cardiovascular mortality vs warfarin to a similar degree across the range of CHA2DS2VASc scores (p-int=0.90, 0.96, 0.21, and 0.37, respectively). TTR in warfarin-treated patients was similar and high (median 68-69%) across CHA2DS2VASc scores, whereas edoxaban trough concentration, exogenous anti-FXa activity and %inhibition of endogenous FXa were higher at increasing CHA2DS2VASc scores. Increasing rates of SSE (1.05→2.99%/year) and MB (2.27→4.66%/year) were observed in the warfarin arm as the CHA2DS2VASc score increased. We classified patients based on CHA2DS2VASc score and compared pharmacokinetics (edoxaban concentration), pharmacodynamics (anti-factor Xa with edoxaban, time-in-therapeutic range for warfarin), efficacy (stroke or systemic embolism ), safety (major bleeding, intracranial hemorrhage ), and cardiovascular mortality, for the approved edoxaban regimen vs warfarin. The ENGAGE AF-TIMI 48 trial randomized patients with atrial fibrillation to once-daily edoxaban or warfarin. The efficacy and safety of the oral factor Xa inhibitor edoxaban compared to warfarin stratified by CHA2DS2VASc scores have not been described.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
